Clostridial myositis in horses
This skeletal muscle infection is best managed in a hospital
Clostridial myositis, also known as gas gangrene, malignant edema, or clostridial myonecrosis, is an infection of skeletal muscle by any of several Clostridium species. Pathogenic bacteria within the genus Clostridium are anaerobic, gram-positive rods that can produce spores that are resistant to various environmental and chemical factors.

The key component for clostridial myositis to occur is the creation of a local anaerobic environment within the skeletal muscle, which can occur with tissue injury, along with the presence of spores or bacteria. There is still debate whether these spores may be introduced at the time of injury or reside within the skeletal muscle prior to injury, and both sources may be valid.1,2 Clostridial myositis is reported to occur most frequently secondary to intramuscular injection, particularly of flunixin meglumine; however, it can also occur because of wound contamination, perivascular injection, castration, foaling injuries, or omphalophlebitis.1-3 Rare cases have no history of open injury, and in these cases, previous seeding of the muscle with spores followed by blunt trauma is suspected.2,4
Once the clostridial spores or bacteria have the correct environment for spore germination and bacterial proliferation, toxin production results in severe tissue necrosis, along with systemic toxemia and bacteremia. Malignant edema is when cellulitis is present without myositis, but it may progress to involve the skeletal musculature.1
Disease symptoms
Clinical signs of clostridial myositis are typically seen within 6 to 72 hours of injury or injection.2 Systemic toxemia may develop very quickly after the onset of clinical signs, and animals may be found dead.4 Initial clinical signs include fever, severe lethargy, tachycardia, reluctance to move, and anorexia. The affected site becomes swollen and painful and develops crepitus and subcutaneous edema.1,4 Horses may become recumbent because of systemic toxemia and severe pain.
Diagnosis
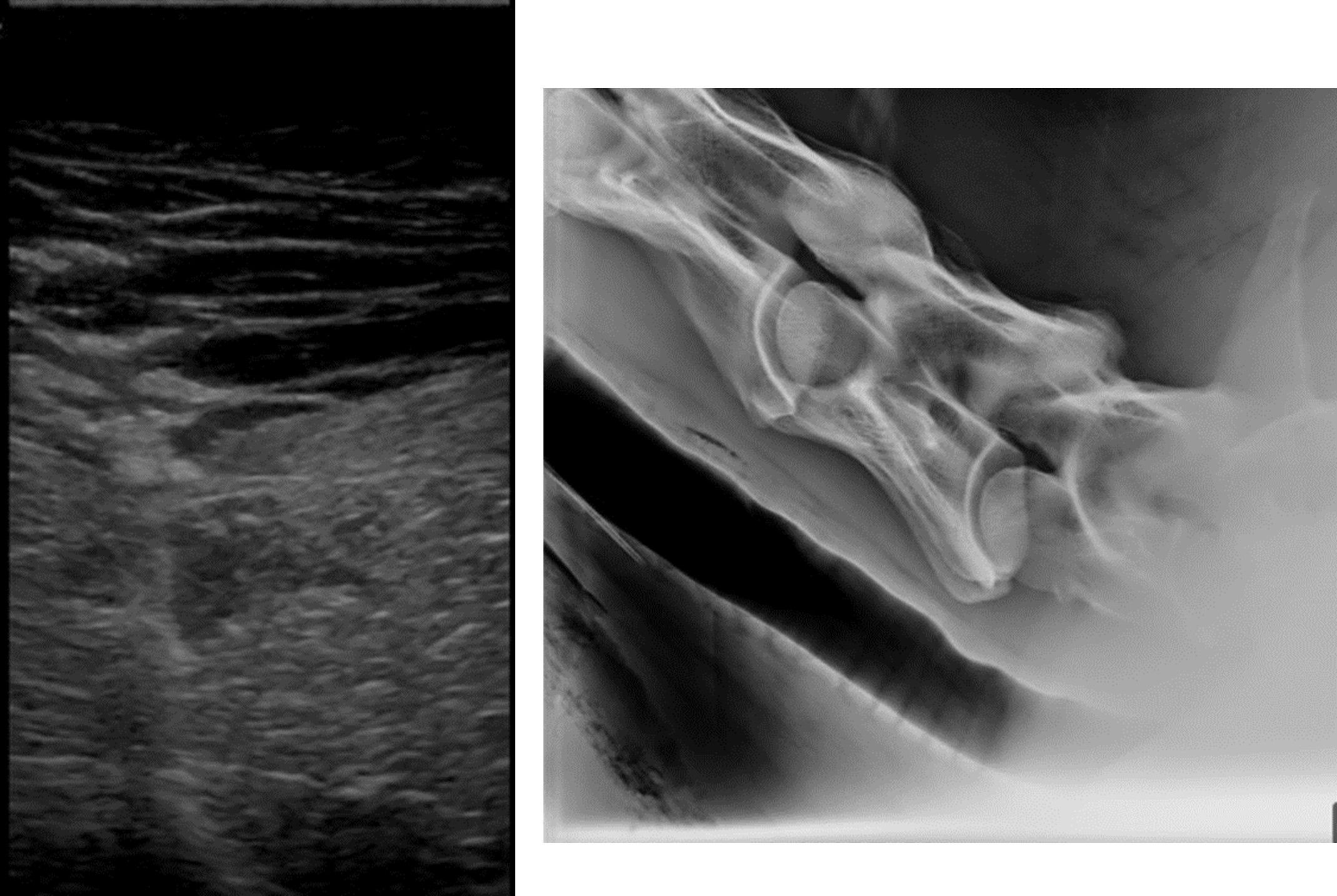
A presumptive diagnosis of clostridial myositis is often made based on clinical signs and history. Aspiration of the swollen, edematous areas reveals serosanguinous, malodorous fluid with or without gas. Gram stain of the aspirated fluid often reveals gram-positive, spore-forming rods.4 Exudate and/or affected muscle tissue can be submitted for fluorescent antibody testing, immunohistochemistry, anaerobic culture, and/ or polymerase chain reaction for confirmation and species distinction, which may affect prognosis.4 Clinicopathologic abnormalities are nonspecific and consistent with a septic process, and elevated muscle enzymes may be noted but often do not correlate with the degree of myonecrosis.1 Ultrasound and/or radiographs of the area will show areas of subcutaneous edema and gas under the skin (Figures 1A and 1B).
Treatment and prognosis
Rapid diagnosis and aggressive treatment are pivotal for the survival of horses with clostridial myositis and, even with appropriate therapy, may be unsuccessful. Surgical fenestration of the skin and fascia, along with debridement of infected muscle tissue, appears crucial to a successful outcome, both through the removal of bacteria and disruption of the anaerobic environment, which lead to bacterial proliferation and toxin production (Figures 2A, 2B, 2C, 2D, 2E and 2F).1,2
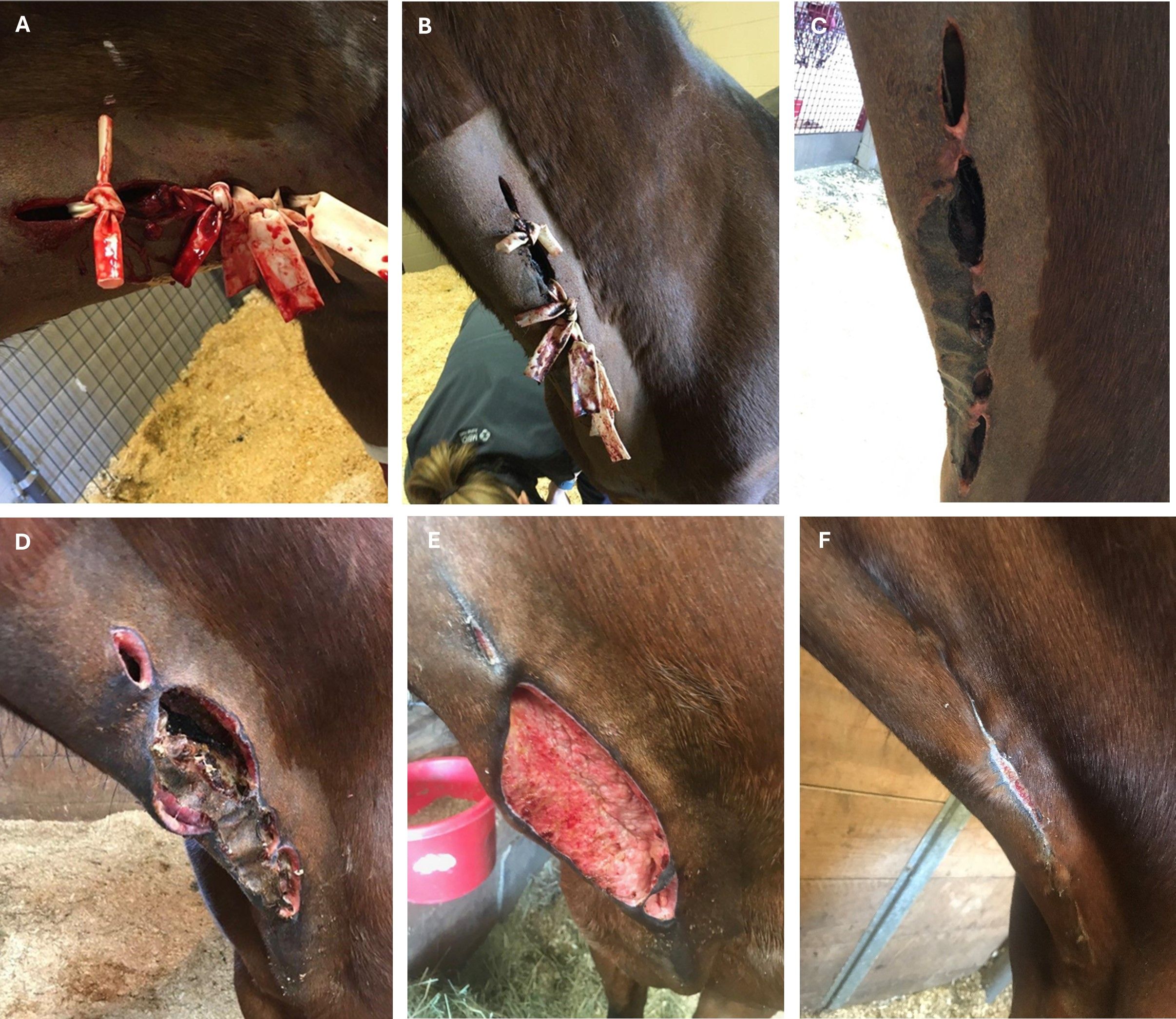
Typically, the skin overlying these areas will necrose and slough, resulting in large wounds requiring long-term therapy. Wound therapy is crucial following fasciotomy and should include continued debridement of infected tissue, hydrotherapy, topical wound care, and additional fasciotomy if needed.
Antimicrobial therapy includes high-dose potassium penicillin (44,000 IU/kg intravenously every 6-8 hours) with or without the addition of metronidazole.1,2 Susceptibility suggests that tetracyclines and chloramphenicol should be effective and may be considered for oral medication options following the discontinuation of intravenous antimicrobials.2 Antimicrobial medications should be continued for a minimum of 7 days following infection resolution.1
Hyperbaric oxygen therapy is often used in humans with gas gangrene and may improve survival when used in conjunction with surgical debridement; however, this is not common equipment in most equine hospital facilities.5 Additional supportive measures may include intravenous fluid therapy, management of anorexia through intravenous or oral routes, and pain management. Additional complications can arise because the primary disease process includes hemolytic anemia2 and can deepen infections.
Complications secondary to systemic toxemia may include disseminated intravascular coagulation, multiorgan failure, and laminitis. Airway management by tracheostomy may be necessary in cases where the neck or head is involved because of severe swelling and edema.
Prognosis is guarded for cases of clostridial myositis, with reported survival rates of 31% to 73%.2,6 Rapid intervention, including fasciotomy, appears key in improving survival rates, with a nearly 40% improvement in survival in horses undergoing fasciotomy in the first 24 hours of hospitalization compared with those with delayed surgical intervention.2 Prognosis is influenced by the Clostridium species present, with C perfringens infections showing the greatest survival, whereas those with C septicum or mixed infections show lower survival rates.2 It is important to remember that therapy must be initiated before identifying Clostridial species for a successful outcome.
Clinical takeaways
Educating owners on the risks of intramuscular injections, particularly of flunixin meglumine, may help reduce the chance of clostridial myositis. These cases are best managed in a hospital setting, and successful treatment requires the owner’s dedication in time and finances. Although this disease is a rare outcome of skeletal muscle trauma, early recognition and therapy are essential for a successful outcome and, even then, can be difficult to attain.
REFERENCES
- MacLeay J. Diseases of the musculoskeletal system. In: Reed SM, Bayly WM, Sellon DC. Equine Internal Medicine. 2nd ed. Elsevier; 2003:461-531.
- Peek SF, Semrad SD, Perkins GA. Clostridial myonecrosis in horses (37 cases 1985-2000). Equine Vet J. 2003;35(1):86-92. doi:10.2746/042516403775467513
- Ortega J, Daft B, Assis RA, et al. Infection of internal umbilical remnant in foals by Clostridium sordellii. Vet Pathol. 2007;44(3):269-275. doi:10.1354/vp.44-3-269
- Uzal FA, Navarro MA, Asin J, Henderson EE. Clostridial diseases of horses: a review. Vaccines (Basel). 2022;10(2):318. doi:10.3390/vaccines10020318
- Wang C, Schwaitzberg S, Berliner E, Zarin DA, Lau J. Hyperbaric oxygen for treating wounds: a systematic review of the literature. Arch Surg. 2003;138(3):272-280. doi:10.1001/ archsurg.138.3.272
- Jeanes LV, Magdesian KG, Madigan JE, Meagher D. Clostridial myonecrosis in horses. Comp Cont Educ Prac Vet. 2001;23:577-587. http://assets.prod.vetlearn.com.s3.amazonaws.com/mmah/f2/68cc5be7ac420399a2bfe1244bbfe4/ filePV_23_06_577.pdf